 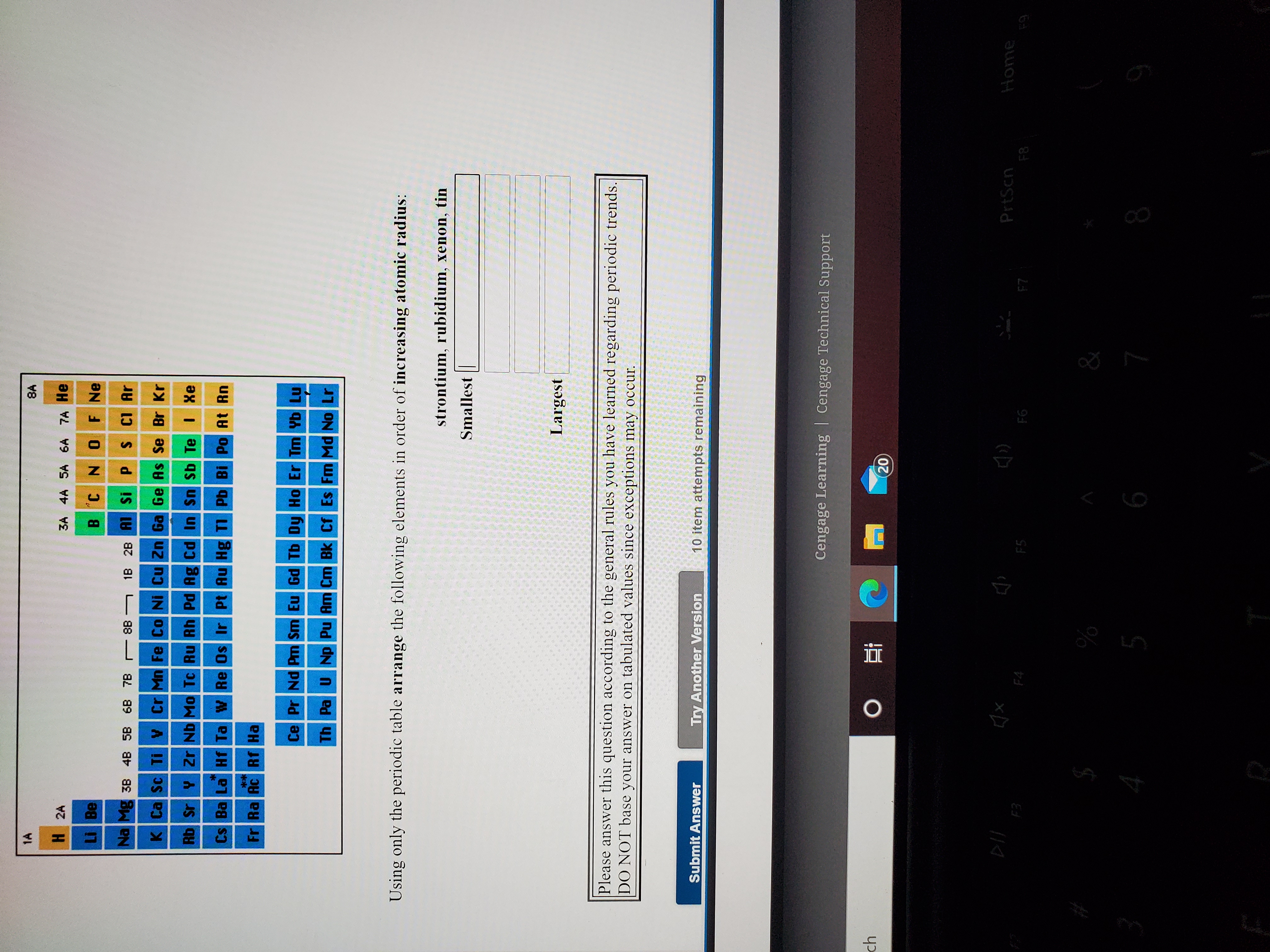
This means that #"Al"#'s atomic radius will be smaller than that of #"K"#, but bigger than that of #"C"# and #"O"#. Largest radius Smallest radius Answer Bank Li Lit Li Arrange the atom and ions from largest to smallest radius. Largest radius Smallest radius Answer Bank Be Mg Mg Rb Ca Arrange the atom and ions according to radius. Another reason for the increase in atomic radius is the electron shielding effect, which states that the electrons in the higher energy levels are being shielded by those closer to the nucleus, further reducing the nucleus' pull on them. Largest Smallest Answer Bank oxygen silicon calcium barium Arrange these elements according to atomic radius. This happens because electrons are now being added to higher energy levels, further away from the nucleus, which weakens the nucleus' pull. As you move down a colomn (a group) of the periodic table, atomic radius increases. 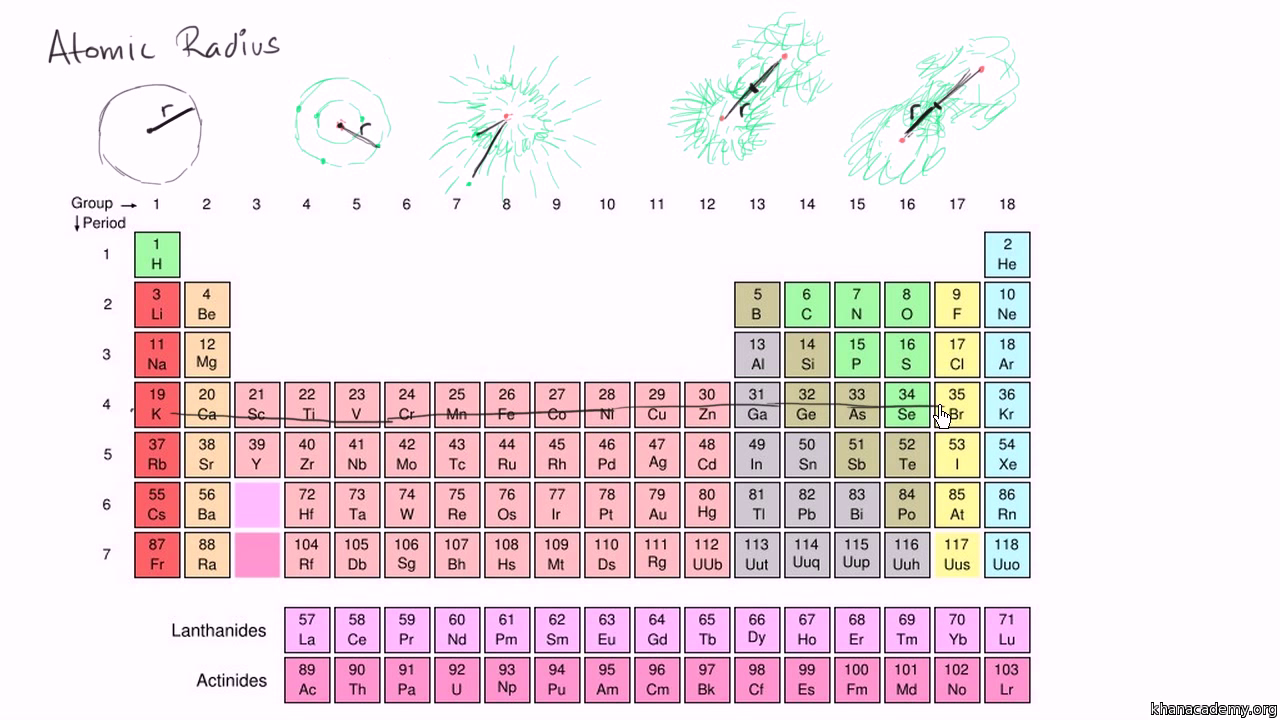 
This is where the other periodic trends comes into play. Since #"C"# comes before #"O"# in the second period, #"O"# will have a smaller atomic radius than #"C"#.Īs mentioned before, #"Al"# and #"K"# don't share a period. We know that atomic radius decreases from left to right within a period due to the increase in effective nuclear charge.Īs you move to the right within a period, the number of protons elements have increases simultaneously with the number of electrons however, electrons are being added to the same energy level across a period, which means that the increasing number of protons allows the nucleus to exert more pull on these electrons, which in turn makes the atomic radius smaller. This aspect will allow you to easily rank them with respect to increasing atomic radius, since both of the periodic trends in atomic size are on display here. The effective nuclear charge on an electron is given by the following equation: Z e Z - S. This site offers comprehensive information for each element including: who, when & where up to 40 properties (chemical & physical) over 3,600 nuclides (isotopes) over 4,400 nuclide decay modes the element names in 10 different languages and more. Elements in the same group have the same number of valence electrons. Elements in the same period (Horizontal) have the same number of valance electrons. The correct order with respect to increasing atomic radius isĪn important thing to notice about these four elements is the fact that only two of them, #"C"# and #"O"#, are in the same row (the same period) of the periodic table the other two, #"Al"# and #"K"#, don't share a period neither with #"C"# and #O#, nor with each other, as #"Al"# is in the third period and #"K"# is in the fourth. The radius of atoms is measured in a picometer. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
